| Papers and Posters | Site Home Page |
PHOTOSYNTHESIS AND STRESS
I.S. Zulfugarov
Institute of Botany, Patamdar Shosse 40, Baku 370073, Azerbaijan, E-mail: zulfugarov@netscape.net
ABSTRACT
In the present study we used wheat (Triticum durum L. and Triticum aestivum L.) leaves and intact chloroplasts and intact algae (Dunaliella salina) cells and measured changes in fluorescence parameters during and after environmental stress treatment. In the present work we have used delayed chlorophyll fluorescence (DF), fluorescence induction kinetics, fluorescence temperature curve (FTC) and other fluorescence technique to investigate acclimation and adaptation processes in plants and algae. Wide range of tolerance to water stress of aboriginal as well as introduced wheat genotypes from various world regions is revealed. Genotypes of Triticum durum L. are differed by greater tolerance to water stress, than ones of Triticum aestivum L. The changes in chlorophyll fluorescence induction kinetics found in wheat are in good agreement with data found for other species. These results suggest that delayed fluorescence (DF) could be used for the screening of plants for drought tolerance. Algae cells were grown under 0.5 – 4 M NaCl conditions. All chlorophyll fluorescence parameters and chlorophyll ratios including the value of the stress adaptation index also declined during recovery at stress conditions. The results of our non-destructive in vivo chlorophyll fluorescence measurements indicate that the mechanism of damage as well as acclimation responses are identical in higher plants and algae.
INTRODUCTION
It is well known that photosynthetic systems in higher plants are most sensitive to high temperature, water and salt treatment [1]. The effects of water, temperature and salt stress on photosynthesis have been a subject of controversy among plant physiologist for many years, and conflicting results have been reported depending on the plant material, and the experimental procedures used for investigations [2]. However, it has not been well established that where and how the chloroplasts are damaged by the treatment and how the damages can be detected easily. Chlorophyll (Chl) fluorescence is one of the few physiological parameters that have been shown to correlate with thermo- and salinity tolerance [3]. In response to a water stress, a decrease in net CO2 assimilaton is generally observed. This effect can however result from different events, such as an inhibition of electron transport activity limiting the generation of reducing power or a limitation in the metabolic activity. Classical method, based on CO2 and water exchange measurements, supply information concerning net photosynthetic activity. However, these informations are not sufficient and additional techniques are required to determine without ambiguity the effects of water stress on photosynthesis.
MATERIALS AND METHODS
Plant culture. For most experiments four hard wheat (Triticum durum L.) genotypes: Garagylchyg-2(drought sensitive), Vugar and Alinja (drought tolerant), Barakatli (frost tolerant) and three soft wheat (Triticum aestivum L.) genotypes: Giymatli-2/17 (drought sensitive), Besostaya (drought sensitive) and Saratovskaya-29 (drought tolerant) were used. Plants were grown at Absheron Basic Field Experimental Station near Baku both under irrigation and drought conditions. Plants for analysis were removed from the field along a soil drying episode in spring beginning from earring to grain ripening.
Algae cells. Dunaliella salina cells were beforehand adapted to medium with 0,5 and 2 M NaCl and then were transferred to concentration of NaCl 0,5; 1; 2; 4 M accordingly.
Chlorophyll Fluorescence Measurements. Fluorescence temperature curves (FTC) defined as a temperature dependence of Chl fluorescence intensity during linear heating (3° C/min) of leaves and isolated chloroplasts under weak light excitation [4] and delayed fluorescence (DF) kinetics [5] are registered. Intensity of fluorescence is registered by Hitachi-850 Fluorescence Spectrofotometer. The experimental set up for delayed fluorescence (DF) was constructed by a phosphoroscope principle [6]. Leaves were kept in the dark for 30 min before measurement of DF. DF was excited with a white light and recorder at 685 nm at room temperature (20° C). Each sample was analysed in 6-8 replications. Wheat leaves were excised under water and placed in vials containing NaCl and MgCl2 solutions of different concentrations.
RESULTS AND DISCUSSION
The influence of high temperature even in the course of short time results in structural and functional changes of photosynthetic apparatus of higher plants and algaes. Temperature also exerts influence on correlation the ways of photosynthetic formation of carbohydrates and their total content [9].
Figure 1 shows a typical traces of the chlorophyll DF induction kinetics of a dark-adapted leaves of wheat genotypes grown both under irrigated and drought conditions. The characteristic phases are clearly resolved. The fast induction kinetics is mainly related to primary photochemistry of photosystem II, whereas the slow kinetics is quite complex and related to interactions between processes in the thylakoid and in the reductive carbon cycle of the stroma [5]. The effect of drought on wheat plants are estimated by the ratio values of maximal (Im) to stationary (Ist) DF intensity Im-Ist/Ist [3] (Figure 2).

Figure 1. Delayed
fluorescence induction kinetics of wheat seedlings under high temperature treatment
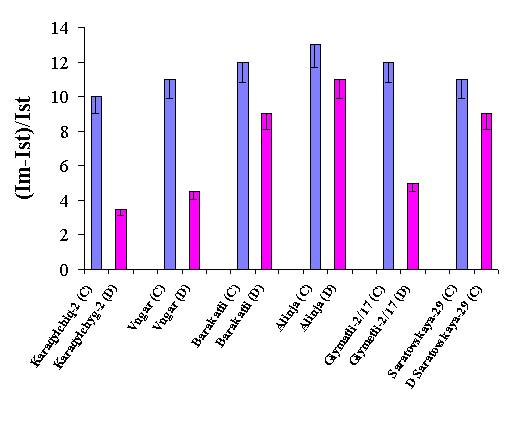
Figure 2. The effect of
drought on delayed fluorescence parameters in wheat plants
Our results showed that when plants leaves are subjected to water stress the slow DF kinetics is markedly affected for drought sensitive cultivars Garagylchyg-2, Besostaya and Giymatli- 2/17 (see Figure 2 and Figure 3).
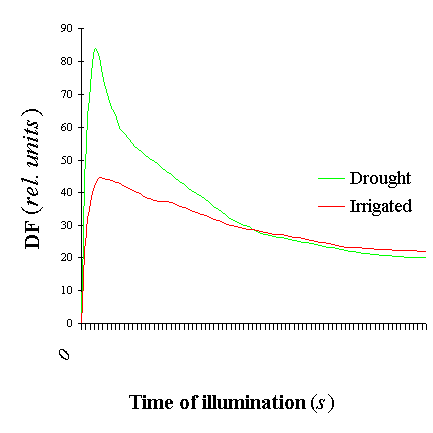
Figure 3. DF induction
kinetics of wheat leaves grown under irrigated and drought condition and subjected to high
temperature (55˚C)
These values are greater for wheat cultivars Alinja and
Barakatli. These data on DF are in good agreement with data on grain yield (Our
unpublished data). Drought tolerant Alinjalost about 30% of grain yield under
drought, whereas drought sensitive Garagylchyg-2 lost more than 60% of yield.
Figure 3 shows the chlorophyll DF induction kinetics of a dark-adapted leaves of wheat
genotypes grown both under irrigated and drought conditions and subjected to high
temperature treatment. As shown in Figure 3, simultaneous treatment of drought and high
temperatures protects the photosynthetic activity against high temperature injury. These
results suggest that delayed fluorescence could be used for the screening of wheat
genotypes for drought tolerance.
When wheat leaves were exposed to increasing temperatures,
the fluorescence intensity started to increase at around 37° C
and two characteristic peaks of higher fluorescence intensity are observed within FTC (Figure
4). The first peak located near (45° C) accompanied by
the loss of PS II activity is apparently associated with changes in the distribution of
excitation energy between the two photosystems. These changes are partly reversible under
transfer the leaves and chloroplasts to low temperatures (Figure 5). This threshold
temperature could be useful for determining relative heat tolerance in different wheat
cultivars.
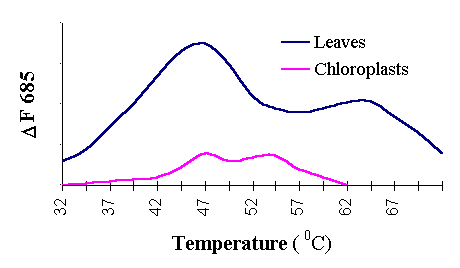
Figure 4. FTC of wheat
leaves and chloroplasts
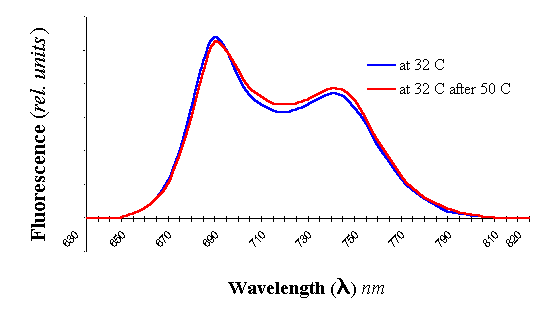
Figure 5. Reversible
changes in fluorescence intensity of wheat leaves under high temperature treatment
The second peak situated above 62° C and has irreversible character (See Figure 4 and Figure 6).
The origin of the second peak is apparently due to thermally induced phase changes in
lipids and proteins and with transition of thylakoids to condensed state.

Figure 6. Irreversible
changes in fluorescence intensity of wheat leaves under high temperature treatment
The treatment of wheat leaves and isolated chloroplasts
with 150 mM NaCl led to increase in the peak intensity, suggesting that such a treatment
induced the physical separation of antenna complexes from PS II, while the treatment with
150 mM MgCl2, led to stabilisation of antenna complexes from PS II and PS I.
First times adaptation (acclimatisation period, 1-2 days)
was conditioned with considerable changes in the induction fluorescence and emission
fluorescence spectra (Figure 7). This period just coincides, now and then, with
stress proteins synthesis. But then, period of proper adaptation comes, which is
accompanied by partial or full restoration of induction fluorescence kinetics and
stationary fluorescence. With 0,5 M NaCl, after 24 hours PS I fluorescence increases and
PS II fluorescence is absolutely absent, which is apparently connected with more labile
structure of PS II. We suppose, that in this time the mechanism of cyclic
photophosporylation starts up for satisfying cells with necessary organic phosphorous,
which after that is done away for protein phosphorylation. Surprisingly, plants and algae
cells acclimated to stress were fully resistant to inhibition of PS I and did not exhibit
any measurable changes at the level of PS I heterodimer [8] abundance and intersystem e-
pool size, although PS II photochemistry was reduced to 66% and 64% respectively.
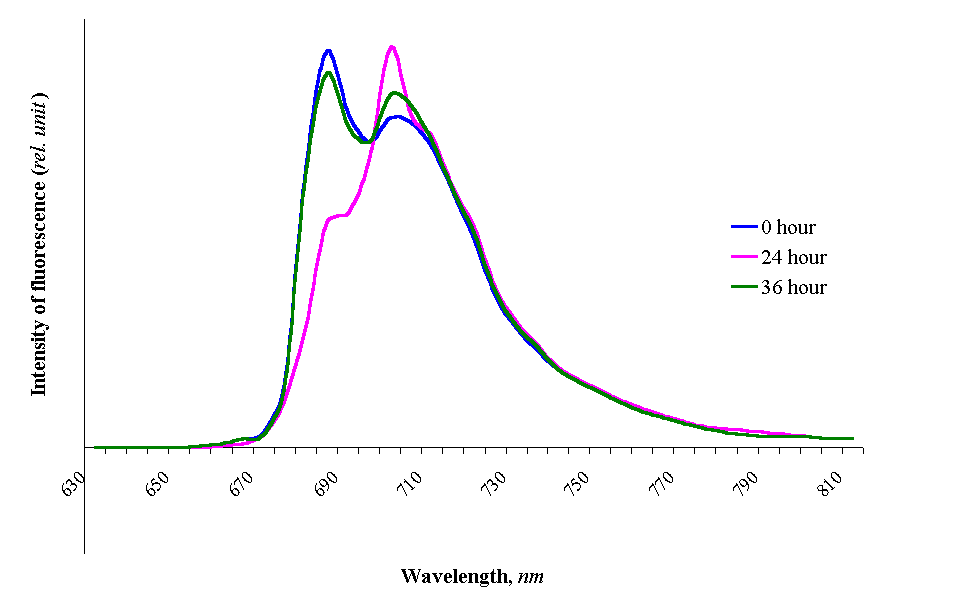
Figure 7. Adaptive
changes in the Dunaliella salina cells adapted to 0,5 M NaCl and then transferred
to 2 M NaCl
The influence of high temperature (37°–70°C), salt (150 – 300 mM NaCl) and drought stress on the functional state of the photosynthetic apparatus and its ability to partially acclimate to these stress conditions was investigated using different parameters of the in vivo chlorophyll fluorescence of whole plants, intact chloroplasts and algae cells. The loss of PS II activity is associated with changes in the distribution of excitation energy between the two photosystems and partly reversible under transfer the leaves and chloroplasts to low temperatures. The treatment of chloroplasts with 0.15 M NaCI led to increase in the peak intensity, suggesting that such a treatment induced the physical separation of antenna complexes from PS II. No differences were detected in the steady-state fluorescence parameters and rapid fluorescence induction kinetics in drought-stressed leaves, indicating that PS II was highly resistant to drought stress alone. However the temperature was above 45˚C, the thermostability of PS II was strongly enhanced in drought-stressed leaves, which was reflected in a smaller decrease in maximum efficiency of PS II photochemistry, coefficients of photochemical and non-photochemical quenching, and efficiency of exitation capture by open PS II reaction centers, and in a smaller increase in the proportion of the QB-non-reducing PS II centers in drought-stressed leaves than in control leaves. This increased thermostability in drought-stressed leaves exposed to high temperature seemed to be independent of the imposed water support since there were no significant variations in the above fluorescence parameters among the drought-stressed plants treated with different water support concentration.
CONCLUSION
Key role in mobilization or formation of
common system of resistance of cells of higher plants and algaes to stress, obviously
belongs to reception systems and transduction external signal. In general, the process of
protein phosphorylation, performing regulator function in response of plants to stress
factors influence is one of important elements of these systems [9]. But the process of
phosphorylation strike up structural changes of photosynthetic apparatus, at the same
time, functional changes of central site in induction of plant response to stress also
leads to phosphorylation/re-phosphorylation of transcriptional factors of stress elements,
perceiving temperature signal and assuming an ability to be connected with promotor zones
of genes and activate their work [10].
Proceeding from these data, it is possible to speak with
confidence, that formation of resistance in strengthen temperature is really connected
with activity of protein-synthesizing system. Such a conclusion quite confirms also to
modern conception of the role of stress proteins in adaptive reaction of plants, being in
the condition of super-optimum temperature, salt, etc. It was determined, that a plant
which was exposed to the effect of high temperature, by inducing the formation of stress
proteins in cells is capable to stand the temperature loading lethal for control (not
exposed to high-temperature effect) plants. It should be noted, that plants of more
heat-tolerant wheat varieties distinguish from un-resistant varieties with the presence of
low-molecular stress proteins, which attribute an adaptive role [11].
In our opinion, the adaptation mechanism of stress factors
(temperature, water deficit, salt, mechanical, osmotic pressure, as well as some
pathogenes), answers to their stress factors as well as process of adaptation of higher
plants and algaes have much in common with these factors. First, most of stress factors
with this or other way tightens osmotic pressure, it tightens structural changes in cell
membrane. Then it takes place protein phosphorylation and signal transduction to genome
and stress protein of so called osmolytes (saccharose, glycerine, betain, prolin etc.) are
synthesized, which is used in adaptive reaction of plants and algaes.
Hence we conclude that fluorescence parameters give an
good experimental technique for investigation influence of the environmental stress
factors in higher plants and algae.
ACKNOWLEDGMENTS
We thank Dr. M.A. Ismailov and D.R. Alieva for their assistance with experiments. We gratefully acknowledge Prof. Jalal A. Aliev for his comprehensive help.
REFERENCES
[1] Falk, S., Maxwell, D.P., Laudenbach, D.E. and Huner, N.P.A. (1996). In Advances in Photosynthesis, V.5, Photosynthesis and the Environment (Neil R. Baker, ed), pp. 367-385. Kluwer Academic Publishers, Dordrecht/Boston/London.
[2] Cornic, G & Massacci, A. (1996). In Advances in Photosynthesis, V.5, Photosynthesis and the Environment (Neil R. Baker, ed), pp. 347-366. Kluwer Academic Publishers, Dordrecht/Boston/London.
[3] Belkhodja, R., Morales, F., Abadia, A., Gomez-Aparizi, J. & Abadia, J. (1994). Plant Physiol. 104: 667-673
[4] Zulfugarov, I.S., Ismailov, M.A. and Aliev, J.A. (1998) In Proceed. 9th International Wheat Genetic Symposium. Saskatoon, Canada. 4:79
[5] Krause, G.H. & Weis, E. (1984). Photosyn. Res. 5: 139-157
[6] Bose, S. (1982) Photochem Photobiol. 36:725
[7] P’yankov V.I., Vas’kovskii, M.D. (1994) Russian J. Plant Physiol. 41:517
[8] Ivanov, A.G., Morgan, R.M., Gray, G.R., Velitchkova, M.Y., Huner, N.P. (1998) FEBS Lett. 430:288
[9] Raz, V., Fluhr, R. (1993) Plant Cell. 5: 523
[10] Gurley, W.B., Key, J.L. (1991) 36:117
[11] Krishnan, M., Nguyen, N.T., Burke, J.J.
(1989) Plant Physiol. 11: 145